If you take supplements that include amino acids in their ingredients, you’re used to seeing amino acids with names like L-tryptophan, L-lysine, and L-arginine. If you’ve spent any time reading up on amino acids, you’ve probably come across some of these same amino acids that are prefaced with a D- rather than an L-. Yet you never see supplements that include amino acids such as D-tryptophan, D-lysine, and D-arginine. This post explains why that is.
Contents
Molecular Structure of Amino Acids
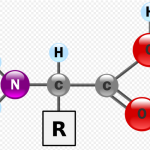
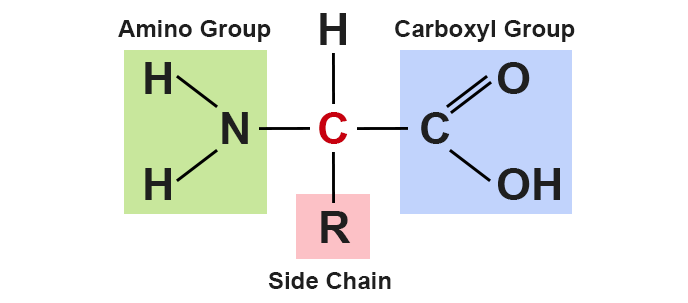
There are dozens of different amino acids, and each is structurally similar (though not identical). Each is a molecule that consist of an alpha, or central, carbon atom linked to four components: a carboxyl group, an amino group, a hydrogen atom, and a variable group (also referred to as an R group, or side chain).
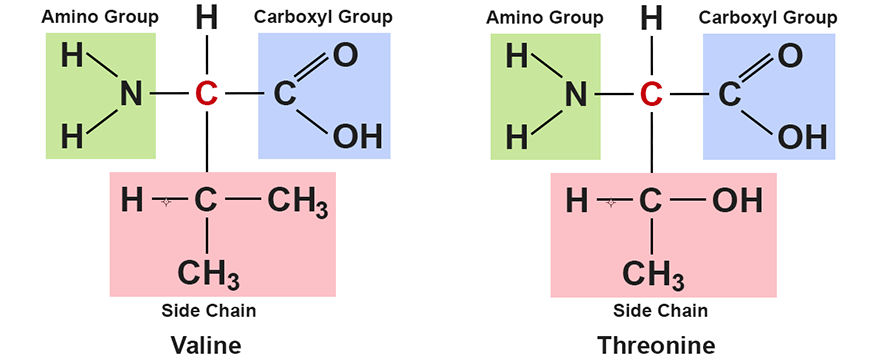
Each amino acid is structurally the same with the exception of the side group. It is the variations in the chemical structures of the side groups that make amino acids differ from one another, and have different properties from one another. This figure shows the molecular structure of two amino acids — valine and threonine. It illustrates that it is only the make up of the side group that differs between the amino acids.
Amino Acids Are Stereoisomers
An isomer is a compound that exists in two different molecular structures, but with the same chemical formula. Each amino acid is an isomer. There are two forms of each amino acid, each with the same atoms, but the atoms are arranged differently.
In addition to being isomers, amino acids are a particular kind of isomer — each amino acid is a stereoisomer. Stereoisomers are mirror images of one another. An analogy might be your hands — your left hand and your right hand each consist of the same “things” (four fingers and a thumb), but they’re mirror images of one another. Which brings us to the L- and D- naming of amino acids.
In the case of amino acids, the name of one form is prefaced with an L- and the name of the other is prefaced with a D-. The L stands for left and the D stands for dexer (which is Latin for situated on the right).
The left and right refers to “the direction in which the crystalline forms rotate polarized light,” which is a definition beyond the scope of this article. There are different ways to represent molecules pictorially, and it’s not always obvious from pictorial representations of the stereoisomers molecules which is left and which is right. The details aren’t critical here — just understanding that both forms of an amino acid are chemically the same but structured a little differently should suffice.
As an example, the amino acid alanine has a molecular formula of C3H7NO2. It’s the same formula for both L-alanine and D-alanine, but when illustrating both stereoisomers you see a difference.
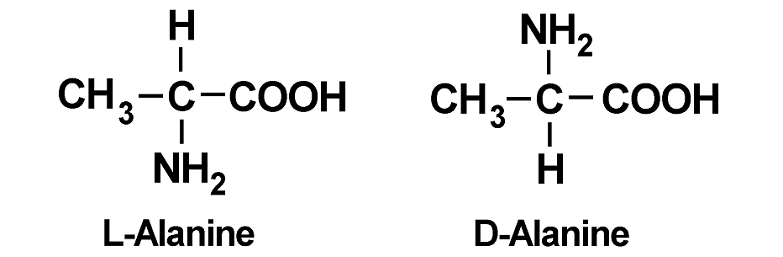
Why L-Amino Acids Win Over D-Amino Acids
D-amino acids are prevalent in bacteria. They aren’t abundant in humans, or any other mammals. The D- form of the amino acid serine is present in human saliva, and may act as a neurotransmitter. All told, though, humans have, and need, very little of any of the D-amino acids.
If the L- and D- forms of an amino acid are chemically so similar, why does the human body prefer the L- form? In a word, the main reason is protein. L-amino acids can be chained together to form proteins. D-amino acids can’t form proteins.
All living things — animal, plant, or single-celled life form — need some proteins. Simple organisms may need protein only for their cell membrane, while higher-level organisms such as humans need it for just about everything. For humans, proteins are considered the building blocks of life. And amino acids are the building blocks of proteins.
Just about all cell functions require proteins. Some proteins act as antibodies, some function as enzymes, and others are important to structural support.
The molecular structural difference between L- and D- amino acids may seem slight, but the differences are significant enough that only the L- forms are capable of being used in protein synthesis.
Proteins can be formed from L-amino acids. Proteins can not be formed from D-amino acids. Not naturally anyway. Researchers have been successful in synthesizing some proteins from D-amino acids, but the body can’t synthesize proteins from D-amino acids. And for us humans, it’s important what the body can do, not what can take place in a laboratory.
Amino Acid Supplements
D-amino acids can’t be used to create proteins, the building blocks of life. They don’t play major roles in human health. That’s why all the amino acid supplements, including anti-aging amino acid supplements, consist of one or more L-amino acids.
While some D-amino acids exist in humans, little is known about their role in human physiology. D-serine may be the most researched of the D-amino acids, and even here research raises as many questions as it answers.
D-serine exists in the brain and contributes to activating NMDA receptors. Cell receptors exist on the walls of neurons (brain cells), and allow specific neurotransmitters to “attach” to them in order to pass information to cells.
NMDA receptors contribute to many important processes that take place in the brain. These receptors are also thought to be involved in several brain-related illnesses, so they’re the targets of some prescription drugs. D-serine has been used in a study involving schizophrenia patients, and the results were inconsistent. Additionally, it’s known that excessive NMDA receptor activation can actually damage neurons and may even contribute to the onset of Alzheimer’s disease.
The other D-amino acid that seems to have some importance in humans is D-aspartic acid. While it doesn’t play any role in protein development (as the L-amino acids do), D-aspartic acid does contribute to the release and regulation of certain hormones — testosterone in particular. That’s why you’ll find D-aspartic acid in some bodybuilding supplements as well as in some male libido products.
Amino Acid Supplements Labeling
Now that you know that in the L vs D amino acids battle, the L-amino acids win. Because there are no known, valid, reasons for intentionally consuming unnatural amounts of D-amino acids, it should be clear why all amino acid supplements contain only L-amino acids.
If you read the labeling of supplements containing amino acids you’ll see them listed with the L- preface, or without any preface at all. Whether an amino acid name is listed with or without the leading L-, it’s referring to the L- form of the amino acid. If both L- and D- forms of amino acids were sold as supplements, excluding the L- preface could lead to confusion. Because only L-amino acids are sold, there’s no confusion as to the form of the amino acid.
Why do some supplement manufacturers include the L-, while other’s don’t? It’s purely an arbitrary choice — simply a preference of that manufacturer. The below figure shows the ingredients labels for two amino acid supplements. One supplement lists the amino acids with the L- included, and one doesn’t. It’s safe to assume that all the amino acids that don’t include the L- preface are, in fact, L-amino acids.
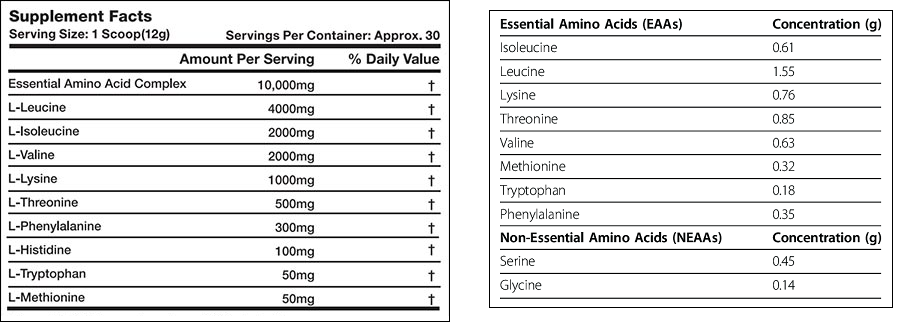
Nootropics stacks — supplements that include several brain-enhancing ingredients in one capsule — often include one or more amino acids. One of the most popular such stacks, Mind Lab Pro by Opti Nutra includes two: L-tyrosine and L-theanine. You can see from the labeling of their supplement they’ve opted to include the L- in the amino acids.
Conclusion
Every amino acid is a stereoisomer — each amino acid has a chemically equivalent version that is molecularly structured as a mirror image. Both versions of one amino acid have the same name, but one name is prefaced with L- while the name of the other is prefaced with D-.
You might be familiar with amino acids such as tryptophan, arginine, and glutamine, and have noticed that they’re often written as L-tryptophan, L-arginine, and L-glutamine. Like every amino acid, there’s also a D- version of these examples: D-tryptophan, D-arginine, and D-glutamine. However, you’ve probably never seen any of these D- prefaced amino acids listed in the ingredients of any supplements. That’s because humans have little need for most D-amino acids.
D-amino acids are abundant in bacteria, but not at all common in humans or other mammals. This is because L-amino acids chain together to synthesize proteins, while D-amino acids don’t have this ability. Proteins are considered the building blocks of life, so amino acids that serve as the basis of proteins are important, while amino acids that play no role in creating proteins are of little value.